How Automation in Cannabis Operations Can Save Time and Reduce Costs

Automation in cannabis operations can streamline a variety of repetitive, time-consuming tasks—from inventory tracking to compliance reporting—helping businesses operate more efficiently and reduce the risk of human error. By automating these tasks, companies can free up valuable time and resources, which can then be focused on growing the business and improving customer experiences.
Reducing Time-Consuming Manual Processes
For many cannabis businesses, managing inventory is one of the most complex and time-consuming tasks. Manual tracking of inventory, product movements, and sales data can result in errors, delays, and inefficiencies. Automation solves this problem by enabling real-time updates of inventory levels, tracking product movements from seed to sale, and generating automatic alerts when stock is low or when products need restocking. This ensures that businesses have an accurate view of their inventory at all times, reducing the likelihood of stockouts or overstocking, both of which can have negative financial implications.
Simplifying Compliance and Reporting
Cannabis businesses are subject to numerous regulations, and staying compliant is essential. However, manually tracking compliance requirements can be overwhelming, especially with the constant changes in laws and regulations. Automation makes compliance easier by automatically generating the necessary reports, tracking product testing, and ensuring all documentation is in order. This not only saves time but also reduces the risk of non-compliance, which can lead to hefty fines or even the loss of business licenses.
Scaling Operations Without the Added Overhead
As businesses grow, manual processes can become bottlenecks that prevent further scaling. Automation helps cannabis companies scale operations without the need for additional staff or resources. Whether it’s automated inventory management, sales reporting, or compliance checks, automation allows businesses to expand without the added overhead of manual systems. This scalability makes it easier for businesses to meet the demands of a growing market while keeping costs under control.
How AirMed Can Help
Our cannabis management software is designed to automate many of the time-consuming tasks that businesses face. From inventory tracking and sales reporting to automated compliance checks, AirMed helps you streamline your operations. By eliminating manual processes, you can not only save time and money but also ensure that you remain compliant with ever-changing regulations. AirMed lets you focus on what matters most—growth and customer satisfaction—while we take care of the back-end operations.
With AirMed, automation is no longer a luxury. By automating routine tasks, AirMed can help your cannabis businesses reduce costs, minimize human error, and scale operations more efficiently. Investing in automation tools is an investment in the future of your business.
Read more about our inventory management here: https://airmedcloud.com/airmed-inventory-management/
Learn about the AirMed automation features here: https://airmedcloud.com/airmed-5-templates/
For more information visit our Software page.
Meeting Health Canada Inspections in 2026

For Canadian licence holders, the “honeymoon phase” of legalization is firmly in the rear-view mirror. Health Canada recently finalized its compliance data for the 2024–2025 fiscal year. With nearly 900 inspections conducted, the data reveals a clear shift in regulatory focus. There was a marked increase in enforcement, with 37 non-compliant reports issued—nearly double the previous year.
What is driving this? While physical security and GAP (Good Agricultural Practices) remain stable, the majority of “Major Observations” now stem from Record Keeping and Data Integrity. Inspectors are no longer just checking if you have a record; they are checking if that record is contemporaneous and attributable.
Common pitfalls identified include the following
Incomplete Batch Records: Gaps in the “narrative” of a batch, specifically around sanitation and nutrient applications.
Traceability Lag: The inability to produce a full “seed-to-sale” history for a specific SKU within a reasonable timeframe during an unannounced visit.
Validation Gaps: Discrepancies between physical inventory and digital logs that suggest a lack of real-time data entry.
Compliance in 2026 is less about avoiding “bad” behaviour and more about proving “good” behaviour through data. As the regulator moves toward a risk-based model, facilities that can demonstrate a “system-first” approach to record-keeping are seeing fewer targeted inspections and smoother license renewals.
Moving forward, achieving a seamless Health Canada inspection requires a shift from reactive data management to a digital-first, continuous compliance model. AirMed software is specifically engineered to provide a unified system of record. Not only does our system let you track every action within the data fields, but we provide bi-directional traceability. And our Custom e-Form Builder provides further traceability, ensuring your facility remains in a state of permanent audit-readiness.
Our system is designed to eliminate the risk of human error and data discrepancies that often trigger regulatory flags. AirMed provides inspectors with immediate, transparent access to your operational history, item-by-item and action-by-action.
To read the Health Canada inspection data, visit: https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/research-data/compliance-enforcement-report-cannabis-inspection-data-summary/2024-2025.html
For more information about Health Canada inspections visit: https://www.canada.ca/en/health-canada/services/cannabis-regulations-licensed-producers/inspections-what-expect-information-package.html
For more information about how AirMed can help, visit our Software page or our Compliance page.
Beyond the Plant: Managing Consumables & Nutrients for True Profitability

In the early days of legalization, the focus for most Canadian producers was simple: “How many grams can we grow?”
But as we move further into 2026, the question has evolved. In a market where quality is now the baseline and price compression is a constant reality, the most successful producers aren’t just tracking their plants—they’re tracking every drop of nutrient and every square inch of substrate.
To truly understand your cost-per-gram, you have to look beyond the plant. Here is how managing your consumables and nutrients with AirMed can turn “hidden costs” into your biggest competitive advantage.
The Hidden Cost of Guesswork
Nutrients, pesticides, and growing media represent one of the largest ongoing expenses in a facility. Yet, many producers still manage these using manual whiteboards or separate spreadsheets. This creates a “data silo” where you know how much you spent on nutrients this month, but you don’t know exactly which batch consumed them.
Preventing the “Friday Afternoon Stockout”
There is nothing more stressful than realizing you are out of a critical bloom booster or beneficial insect treatment on a Friday afternoon. For Micro Producers, who often operate with leaner inventories to preserve cash flow, a stockout can be devastating to a harvest cycle.
AirMed provides real-time visibility into your non-cannabis inventory. This ensures you only “pay for what you need,” keeping your capital focused on growth rather than sitting on a shelf as excess.
Sustainability & Environmental Reporting
Effective nutrient management isn’t just about saving money; it’s about social responsibility. By using AirMed to track your nutrient applications and runoff data, you create a digital trail that proves your commitment to 4R Nutrient Stewardship (Right Source, Right Rate, Right Time, Right Place). When it comes time for environmental reporting or applying for “Craft” or “Organic” certifications, your data is already organized and export-ready.
ROI: Linking Inputs to Outcomes
The ultimate goal of tracking consumables is to find your “sweet spot.” Does that expensive new terpene-enhancer actually result in a higher wholesale price for the final product? By linking your Inventory Ledgers with your nutrient logs, you can run an ROI analysis: Batch A (Standard Nutrients) vs. **Batch B (Premium Additives). Which one yielded a higher profit margin after all input costs were deducted?
Conclusion: Data is the New Fertilizer
In 2026, the producers who thrive will be the ones who treat their nutrients and consumables with the same level of scrutiny as their THC percentages. By integrating your consumable tracking into your AirMed workflow, you gain a 360-degree view of your facility’s health—and your bottom line.
Read more about our inventory management here: https://airmedcloud.com/airmed-inventory-management/
Learn about the AirMed Micro advantage for small producers: airmedmicro.com
For more information visit our Software page.
The Paperless Cannabis Facility: Why 2026 is the Year to Ditch the Binders

For years, the “compliance binder” has been a staple of the Canadian cannabis facility—a growing mountain of paper containing sanitation logs, visitor sign-in sheets, and equipment maintenance records. With Health Canada’s audit processes becoming more data-intensive and the margin for operational error shrinking, the risks of a paper-based system outweigh the perceived simplicity of a clipboard.
At AirMed, we believe that compliance shouldn’t be a chore that halts production. Here is how you can transform your facility into a streamlined, paperless operation using the advanced tools within the AirMed ecosystem.
The Power of the Custom e-Form Builder
One of the most transformative features of AirMed is our Custom e-Form Builder. While we introduced this functionality to help automate workflows, its true value in 2026 lies in total facility digitization.
Instead of manual logs that can be lost, stained, or misfiled, AirMed lets producers create digital versions of essential documents. For example, ensure staff are prompted to complete cleaning tasks at the right intervals, with digital timestamps that prove compliance. You can also track the “health” of your HVAC, irrigation, and lighting systems. And so much more.
By moving to a digital format, you don’t just save paper; you create a searchable database. If an auditor asks for the sanitation history of Room 4 from six months ago, you are a click away from a professional report, rather than a frantic search through a dusty filing cabinet.
Audit Readiness: From “Days of Prep” to “Real-Time”
We’ve often discussed the importance of Data-Driven Decision Making, but in the context of a paperless facility, data is your best defense. A traditional paper system is “reactive”—you only find errors when you review the logs.
AirMed shifts your facility to a “proactive” stance. Because our software is cloud-based and supports fully customizable tables, management can see in real-time which tasks have been completed and which are overdue. This level of transparency is exactly what Health Canada looks for during an inspection.
Scaling for Micros: Professionalism on a Budget
For micro-cultivators, the move to paperless is even more critical. AirMed Micro was purpose-built to provide the same award-winning platform used by Canada’s largest LPs but at a price point that fits a smaller scale. By utilizing the modular add-ons for Quality Management and Custom Forms, a micro producer can operate with the same level of regulatory sophistication as a large enterprise, without the need for a dedicated compliance department.
Integrating the Whole Operation
A paperless facility isn’t just about the grow rooms. It’s about how that data flows through the rest of your business. As we highlighted in our 2025 updates, AirMed’s integration with tools such as QuickBooks ensures that your records match your production perfectly. With our Inventory Ledgers and intelligent backdating, there’s no need to cross-reference a paper log with a digital sales order; the system does the heavy lifting for you.
Conclusion: A Competitive Advantage
In 2026, the most successful Canadian producers—whether large or small—will be those who treat compliance as a digital asset rather than a regulatory burden. Transitioning to a paperless facility with AirMed reduces human error, slashes the time spent on reporting, and ensures that when an auditor knocks, your facility is ready.
For more information visit our Software page.
You can also read a blog post about our form builder here: https://airmedcloud.com/custom-eform-builder-airmed-5/
And learn more about our inventory management here: https://airmedcloud.com/airmed-inventory-management/
Pivoting After Health Canada’s CBD NHP Ruling

Why the Medical Channel is Now the Primary Path for CBD
The start of 2026 has brought a significant shift for the Canadian cannabis industry. In a move that surprised many stakeholders, Health Canada recently removed the long-awaited proposal for Natural Health Products (NHPs) containing CBD from its forward regulatory plan. For years, producers and consumers alike hoped for a “third stream” for CBD—one that would allow non-intoxicating cannabidiol to be sold on pharmacy shelves and in health food stores alongside vitamins and supplements. With that pathway now effectively stalled, the industry must pivot.
Here is what the removal of the CBD NHP proposal means for your business and how you can navigate this “Medical-First” landscape.
What Happened?
On January 8, 2026, industry news outlets reported that the regulatory framework designed to create a “pathway for health products containing cannabidiol” had been stripped from the government’s upcoming agenda. This follows years of consultations that began back in 2019.
Is the Medical Channel the Only Way Forward?
For producers focused on the wellness and therapeutic benefits of CBD, the answer is increasingly “yes.” Without an NHP pathway, CBD remains locked behind the same regulatory requirements as THC.
However, this isn’t necessarily a setback—it’s a clarification of the market. The Medical Cannabis Channel remains the only legal way for producers to provide CBD to patients with professional healthcare oversight and without the “recreational” stigma of the adult-use retail environment.
How AirMed Supports Your Pivot to Medical
If your business strategy relied on CBD NHPs, now is the time to double down on your medical sales infrastructure. AirMed was built from the ground up to support the complex requirements of medical cannabis sales in Canada.
1. The AirMed WordPress Medical Plugin
Your website needs to be more than a digital brochure; it needs to be a secure patient portal. Our WordPress Medical Plugin allows you to:
- Manage Patient Registration: Streamline the intake of medical documents and healthcare practitioner (HCP) authorizations.
- Secure Product Catalogs: Show your CBD-dominant products only to verified medical clients, ensuring you stay compliant with strict promotion and advertising regulations.
2. Encrypted Patient Messaging
Health-focused CBD consumers often have more questions than recreational users. AirMed provides secure, encrypted messaging between your staff and your clients, allowing you to provide high-touch customer service while protecting sensitive patient data.
3. Seamless Fulfillment & Shipping
Transitioning to a direct-to-patient model requires a robust logistics engine. AirMed integrates directly with carriers and fulfillment tools (like ShipStation) to ensure that your wellness-focused clients receive their CBD products discreetly and efficiently.
The Bottom Line
The removal of the CBD NHP proposal from the 2026 regulatory plan is a reminder of the volatility of the Canadian regulatory environment. However, the demand for high-quality, health-focused CBD has never been higher. By leveraging the Medical Channel, producers can bypass the uncertainty of the NHP path and start serving patients today.
Official Context: You can monitor the current Health Canada Forward Regulatory Plan for updates here: https://www.canada.ca/en/health-canada/corporate/about-health-canada/legislation-guidelines/acts-regulations/forward-regulatory-plan.html
Industry Commentary: StratCann reported that this move leaves CBD strictly under the Cannabis Act, rather than the Natural Health Products Regulations: https://stratcann.com/news/health-canada-removes-cbd-nhp-regulation-proposal-from-forward-regulatory-plan/
And for a demonstration of our medical plugin in action, please visit: https://airmeddemo.com/
Budget-friendly Cannabis Management

In a previous post, we announced AirMed Micro, our new budget-friendly cannabis management licensing option.
But why is AirMed the ideal choice for micros? Because you only pay for what you need right now. Modular pricing means you customize the system to your needs and only pay for what you use. The result is software priced significantly lower than other cannabis management systems, making it an ideal choice for micro cultivators operating on a budget.
Built on the award-winning AirMed platform, AirMed Micro lets you utilize and pay for only the modules you need at the time, with the option to add more later. Scale up by adding new features when your business demands such as optional integrations with third-party systems like Quickbooks and Shipstation, which cut costs and consolidate all aspects of your operation in one convenient platform.
This flexibility makes AirMed Micro an investment in the future of your operations. Our pricing structure offers long-term cost savings while still delivering top-tier features.
To learn more about AirMed Micro, please visit our new AirMed Micro website including pricing and free trial information: https://www.airmedmicro.com/
If you have questions not answered on the site, contact our sales department using the Request Demo link at the top of the page. You can also drop us a line at info @ airmed dot ca or call 1-877-313-2442 toll free.
AirMed Introduces Scaled-down Version for Micros

To meet market demands, we’re now offering a new licensing option for micro cultivators that provides an affordable alternative to higher-priced ERPs.
This new version relies on AirMed’s modular design, letting clients license only the modules they need right now. The micro option delivers a scaled-down version designed specifically for small operations.
Micro producers get an intuitive and affordable cannabis management solution that simplifies the complexities of cultivation, inventory management, and compliance. But the system can grow with you. We provide the ability to add functionality when needed without having to switch to a new software system. You can start with the core features and add modules as your production scale and budget increases.
Designed to improve efficiency in smaller operations, AirMed Micro integrates all essential aspects of cannabis cultivation into one user-friendly platform. But we’ve simplified data entry with intuitive graphical interfaces and minimal manual input to reduce overhead as well as the potential for errors.
Our real-time dashboard lets you track everything from source materials to sales orders instantly and helps you make informed decisions to enhance quality and optimize inventory.
Offering a streamlined version of the full AirMed platform, AirMed Micro provides the essential features needed to ensure complete operational control with optional add-ons to suit growing needs.
Easy to use, fully compliant, with fast set up and flexible billing, our new modular licensing makes AirMed ideal for micros on a budget.
To learn more about AirMed Micro, please visit our new AirMed Micro website including pricing and free trial information: https://www.airmedmicro.com/
If you have questions not answered on the site, contact our sales department using the Request Demo link at the top of the page. You can also drop us a line at info @ airmed dot ca or call 1-877-313-2442 toll free.
AirMed & Cannabis Inventory Management

Last week we discussed the benefits of inventory management. Read the post here.
This week, we’ll look at ways that AirMed helps streamline inventory management for cannabis businesses.
In 2024 we introduced ledgers designed to streamline your inventory management and recordkeeping.
AirMed integrates inventory ledgers throughout the system letting you effortlessly track all transactions related to your inventory. Our inventory ledgers provide detailed tracing of every addition or reduction in batches or lots with full audit trails. We also added intelligent backdating to ensure adjustments or entries made in the past won’t create discrepancies in the future. And we’ve made it easy for you to resolve inventory inconsistencies by offering multiple tools for making corrections.
But ledgers are not the only inventory management tools in AirMed.
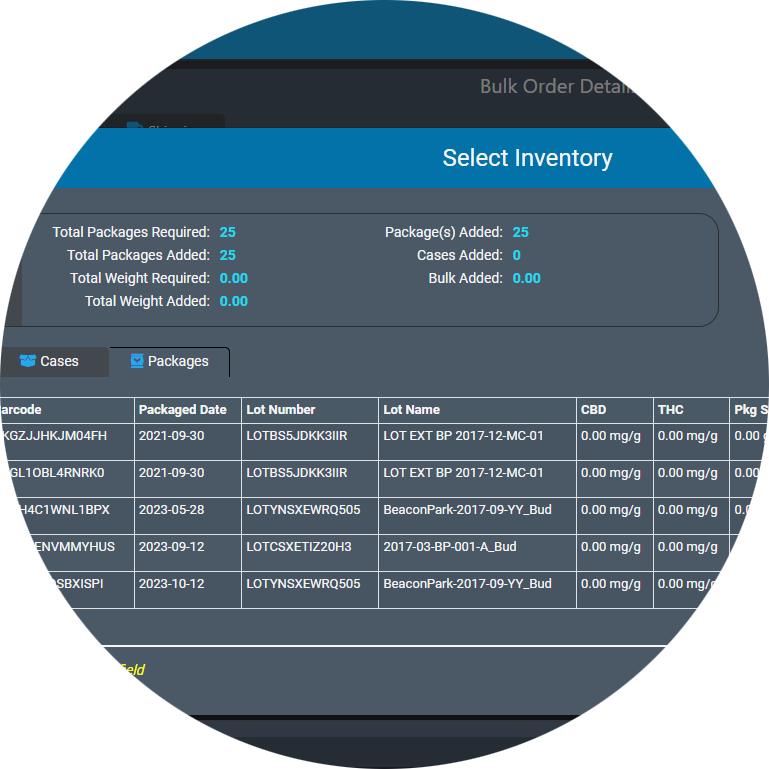
Our software offers GS-1 barcoding with full functionality for multi-level packaging that lets you assemble retail-ready individual packages using a ‘master case’ system and identify & track products at each packaging level.
AirMed offers unparalleled ease while dealing with complex order requirements. Manage order requests, order planning & assembly, as well as shipping from one screen. After releasing a planned order to production, verify order items in one step. You can select available inventory from multiple lots for order fulfillment. Browse products, package sizes and available inventory, then initiate package runs and pack cases workflows in the items table.
Since AirMed reports on every item in the system, inventory reporting is available at the click of a button. Real-time inventory tracking lets businesses assess product demand more effectively, reducing excess inventory and lowering costs associated with unsold goods.
Utilizing cannabis inventory management is more than just a way to keep track of products—it’s a key to reducing losses, improving operational efficiency, and boosting profitability.
AirMed has been 100-percent Canadian owned and operated since it was created in 2014. Click the Request Demo button at the top of the page today to explore AirMed in a free walkthrough and learn what home-grown can do for you.
For more information about AirMed visit our Software page.
To read more about our inventory ledgers, read our previous posts.
AirMed 5 Introduces Inventory Ledgers
Data-Driven Decision Making & AirMed
Data-driven decision-making is the practice of using facts, metrics, and data to guide strategic business decisions.
From optimizing cultivation processes to enhancing marketing strategies, the ability to analyze and act on data can make the difference between thriving and struggling in this dynamic cannabis space. By leveraging data, cannabis operators can gain actionable insights, reduce costs, improve yields, ensure compliance, and ultimately achieve a competitive edge.
Benefits of Data-driven Decision Making
Cannabis cultivation is a complex process influenced by numerous variables. By using data analytics, producers can monitor these variables in real-time, identify trends, and make adjustments to maximize yield and quality.
Cannabis is a resource-intensive product. Data-driven strategies help identify inefficiencies and optimize resource allocation. This ensures that resources are used as effectively as possible and reduces waste. These measures not only decrease operational expenses but also support sustainability initiatives—an important consideration for environmentally conscious consumers.
Compliance requires meticulous record-keeping and traceability throughout the supply chain. Data-driven systems that leverage information to demonstrate adherence to quality and safety standards enhance credibility with regulators and customers alike. Moreover, predictive analytics can help identify potential risks, enabling proactive measures to mitigate them.
As the cannabis industry matures, innovation is key to staying ahead of competitors. Data-driven insights can empower you to experiment with new cultivation techniques, product formulations, and market strategies. By staying informed and agile, you can seize opportunities and respond effectively to market shifts.
With narrow profit margins and significant operational costs, data-driven decision-making can also support financial sustainability. Data analytics can provide insights into cost drivers, helping you identify areas where they can reduce expenses or increase efficiency. Analyzing labor costs and production timelines might highlight opportunities to streamline operations, while inventory data can ensure that stock levels align with demand, reducing the risk of overproduction or stockouts.
AirMed & Your Data
AirMed has always gone beyond other systems by tracking and reporting thousands of fields of data.
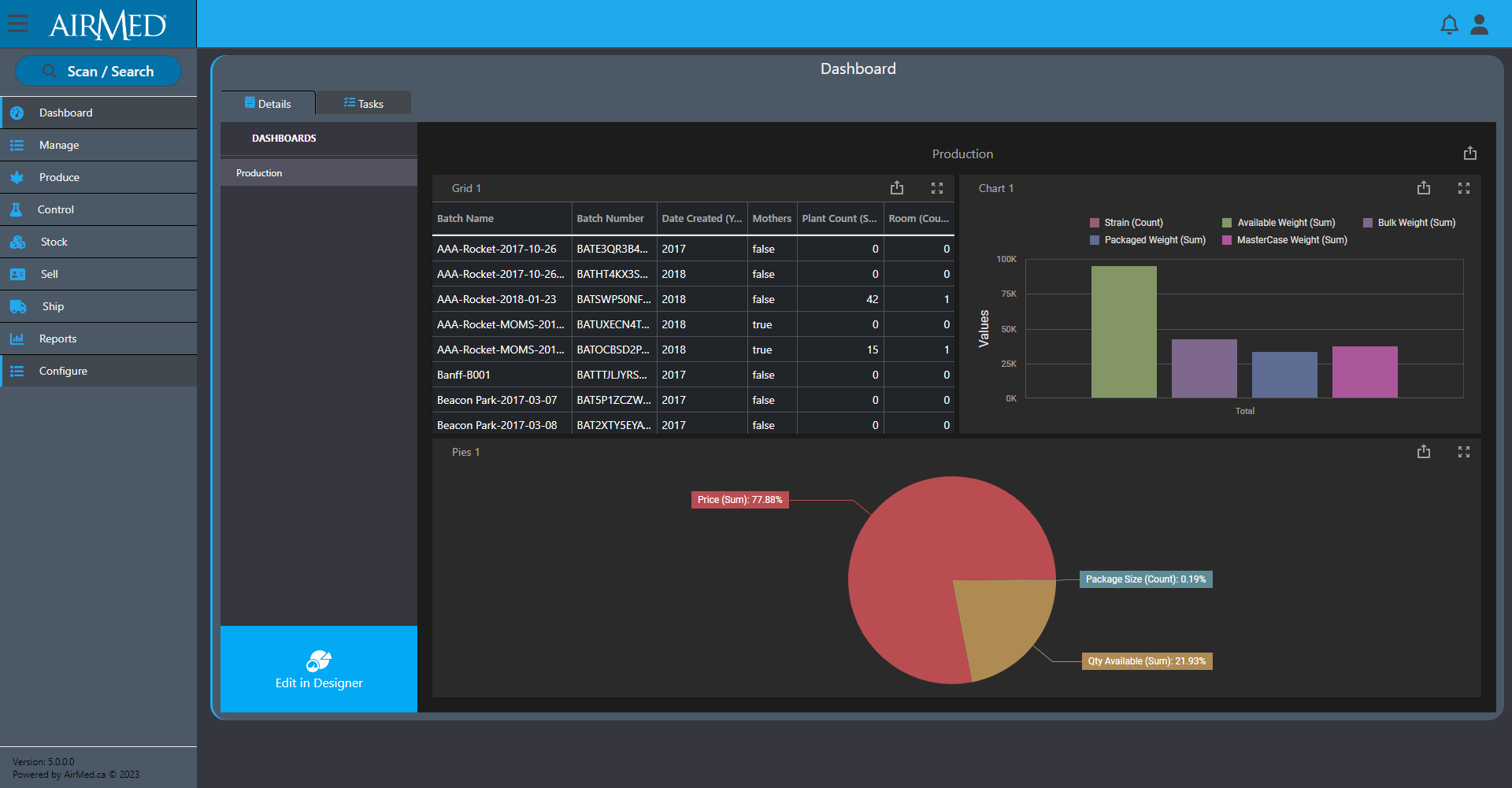
Dozens of reports come standard with AirMed, but we also offer an optional report designer to create unlimited custom reports with access to every field of data in the system. You can select individual fields to include in a report and add options to manage master-detail relationships, cross-tab reports, table and vertical reports, and filter options.
And our optional business intelligence (BI) designer provides pre-designed dashboard widgets that offer the best data visualization option for you. You can create insightful and information-rich decision support systems by simply selecting the appropriate UI widget: Chart, Pivot Table, Data Card, Gauge, TreeMap, Map, Grid, or simple Filter elements. By dropping data fields, results are immediate, accurate and always relevant.
For those who wish to edit or create new widgets, the BI Dashboard is engineered to let you spend more time on business and less on UI customization. Whether it’s manipulation of individual chart series, specifying a pivot table’s dimensions or connecting UI elements to fields across different data-sources or data providers, the BI Dashboard designer is built to make your experience a productive one.
Conclusion
Incorporating data-driven decision-making provides the ability to collect, analyze, and act on data to optimize processes, improve product quality, reduce costs, ensure compliance, and remain competitive. AirMed has been designed to help you embrace a data-driven approach.
For more information visit our Software page.
Inventory Ledgers: Adjustments & Corrections
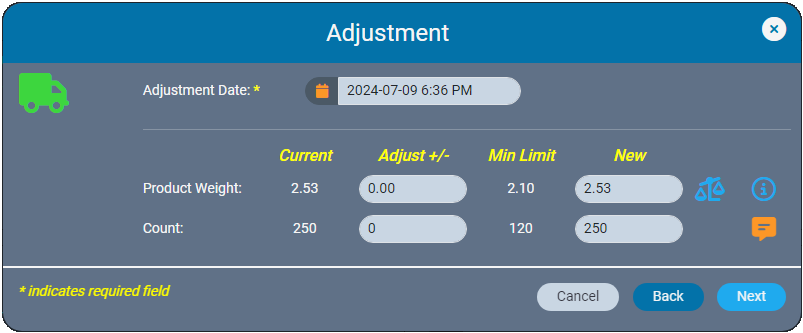
We recently announced the introduction of Inventory Ledgers in AirMed 5. This new functionality was designed to streamline your inventory management and recordkeeping. In this post we go into more details about the adjustments and corrections features in our inventory ledgers.
In the event of data entry errors, inventory ledgers make it easy to adjust weights and counts to resolve discrepancies. AirMed ensures that backdated adjustments are logically aligned with all prior and subsequent records. The ledger prevents negative balances by automatically validating that backdated weights or quantities are consistent with subsequent records.

Whether you are making a correction immediately or at a later date, AirMed offers multiple correction tools. Previously found in the Actions Menu, these options are now located in the ‘Admin Actions’ section, visible only to users with supervisor-level access.
For example, if you create a new inventory item with an incorrect date, you can amend the creation date based on the date of the parent record. All changes are logged in both the affected item’s ledger and the parent record ledger.
Additionally, new options are available to delete incorrect records or merge materials back into their original source when appropriate.
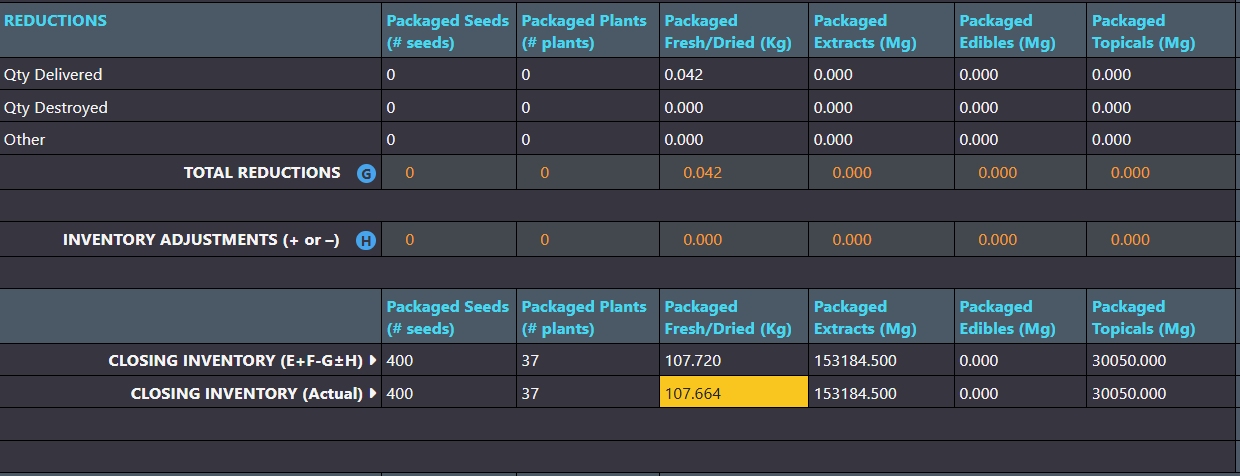
Adjustments that affect inventory levels are clearly categorized in monthly compliance reports.
While mistakes are inevitable, you can leverage AirMed’s technology to get you back on track.
We designed our new inventory ledgers to empower you to correct errors and ensure that your records accurately reflect your physical inventory. For more information about AirMed 5 visit our Software page.
Inventory Ledgers: Intelligent Backdating
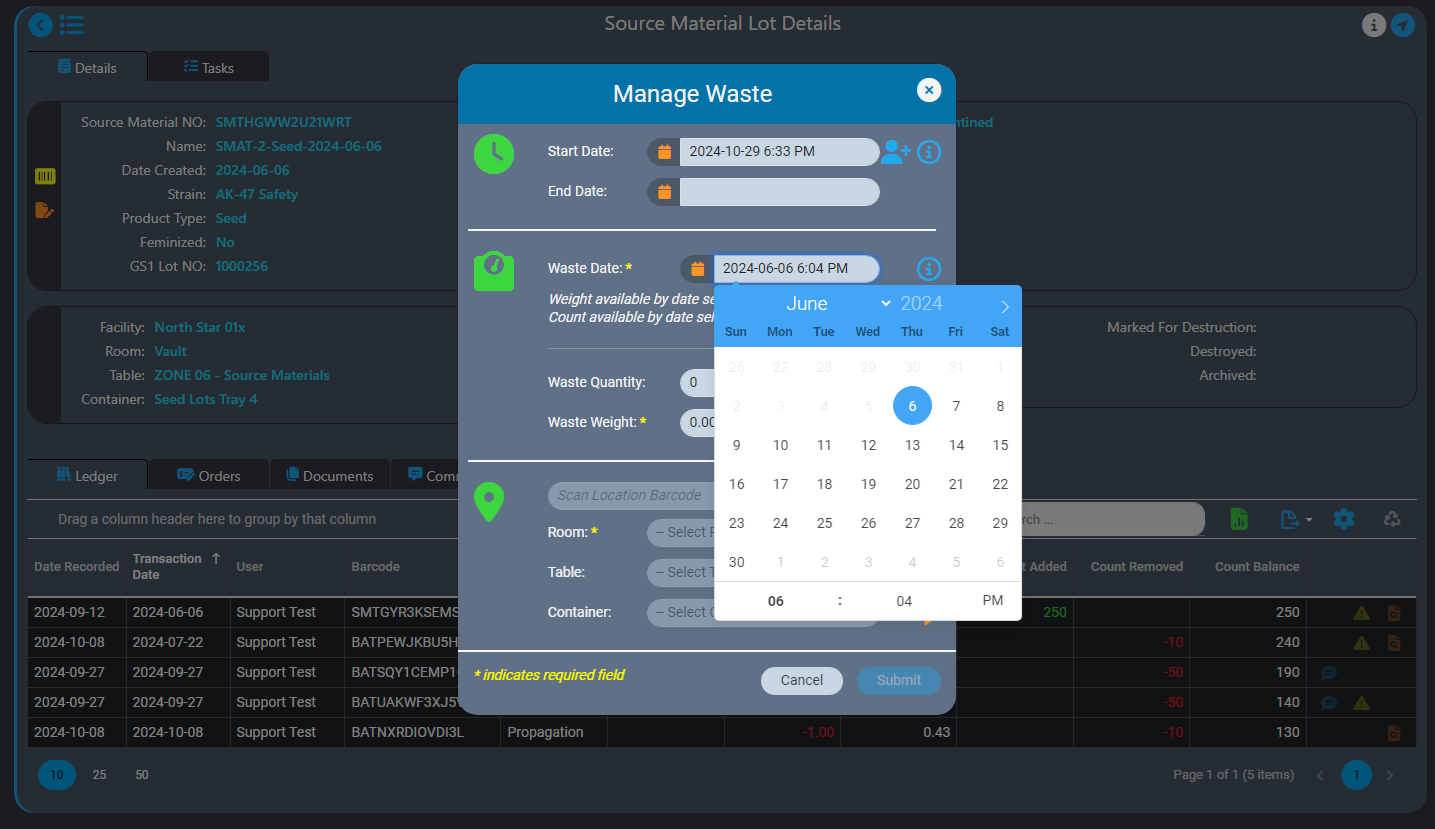
We recently announced the introduction of Inventory Ledgers in AirMed 5. In this post we go into more details about the intelligent backdating feature in our inventory ledgers.
Our intelligent record backdating ensures that any quantity or weight you enter will not lead to discrepancies in subsequent records. When backdating an adjustment, AirMed checks that it aligns logically with all successive records.
For instance, if you need to log waste for a source material, our safeguards won’t let you accidentally backdate a record to a date that is before the source material was created. You can only enter a date that is after the date the source material was created.
If an inventory item is created with the wrong date, admin tools can be used to adjust the creation date to match the creation of the parent record (e.g., adjusting a lot’s creation date to align with the date of the harvest). All changes are logged in both the affected item’s ledger and the parent record’s ledger.
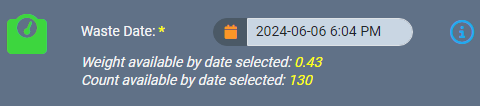
If a backdated action impacts monthly compliance reporting, AirMed will automatically generate an incident within our built-in Quality Management System (QMS). Supervisors can then investigate and resolve the incident, document the findings, and track it through a severity assessment and impact evaluation.
Having an efficient way of tracking and resolving discrepancies not only helps you meet compliance but also saves you time and money.
Intelligent backdating lets you reconcile your data with your physical inventory while satisfying your regulatory obligations. For more information about AirMed 5 visit our Software page.
Inventory Ledgers: Tracking
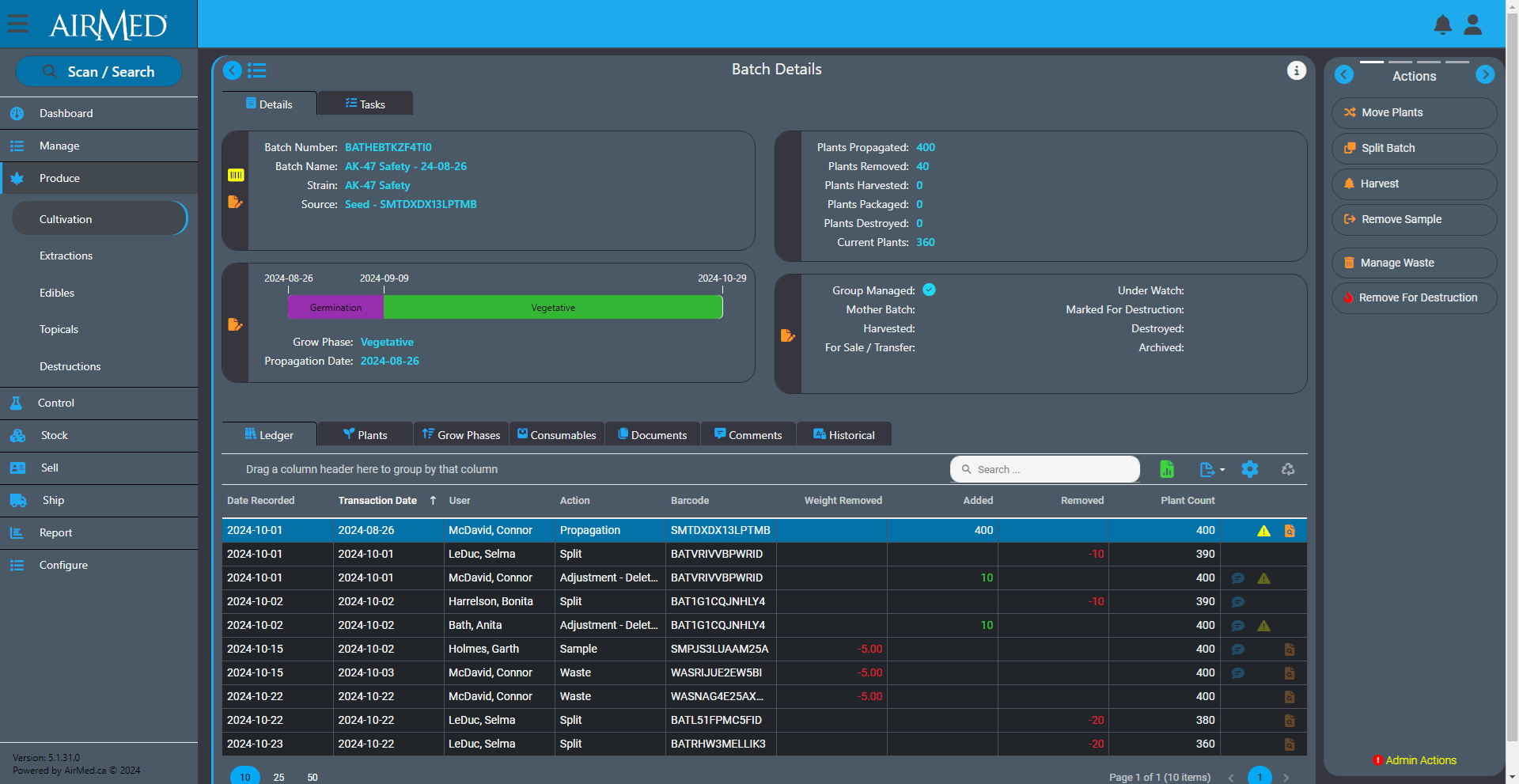
We recently announced the introduction of Inventory Ledgers in AirMed 5. This new functionality was designed to streamline your inventory management and recordkeeping.
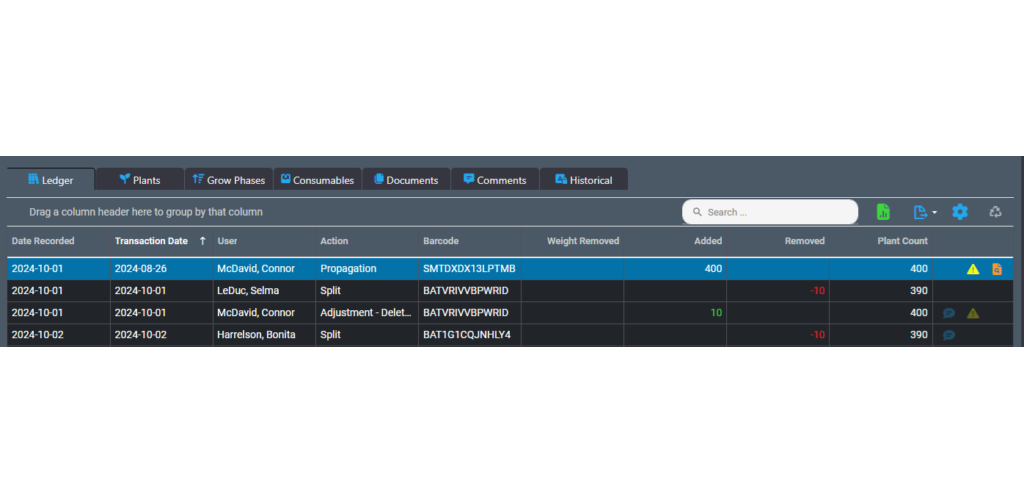
The ledger lets you track actions performed on an inventory item. These actions are referred to as transactions in the ledger. You use the ledger as a perpetual register that records each transaction for an inventory item.
To use the ledger for a batch, for example, open the Batch Details screen and look for the Ledger tab. The tab shows a table that you use to record individual transactions related to that batch.

Each row offers fields for the date that the transaction took place along with the date the transaction was recorded in case they are different. There are also fields for the type of action that occurred, the user who performed the action and weights or counts added or removed during the action. The final column provides icons to visibly indicate adjustments or corrections to the batch. You can re-order the columns in the table by dragging.
The benefits of inventory ledgers are seen in both operational efficiency and compliance. Not only do they let you monitor the progress of a batch or lot, but tracked actions can be used to create a template for future production. During an audit, the ledger serves as a detailed register about each item, which can be used to validate processes and verify physical inventory.
Our inventory ledgers are designed to help you optimize your operations and ultimately enhance profitability. For more information about AirMed 5 visit our Software page.
AirMed 5 Introduces Inventory Ledgers

AirMed 5 now features powerful new functionality to simplify inventory management and improve recordkeeping. With the integration of inventory ledgers throughout the system, you can effortlessly track all transactions related to your inventory.
Comprehensive Tracking
Inventory ledgers provide detailed tracking of every addition or reduction to inventory, whether in batches or lots. Each entry includes the user responsible for the transaction, along with the date, time, transaction type, and the corresponding weight or quantity. This ensures full visibility into the usage of each batch and lot, providing a clear audit trail. Additionally, clickable links enable quick access to related records and associated comments.
Intelligent Backdating

Intelligent record backdating ensures that adjustments or entries made in the past do not cause discrepancies in future records or reporting. For example, if waste for a source material needs to be logged, AirMed ensures that the waste entry can be backdated only after the source material was created. The ledger also prevents negative balances by automatically validating that backdated weights or quantities are consistent with subsequent records. If a backdated action impacts monthly compliance reporting, AirMed will automatically generate an incident within our built-in Quality Management System (QMS).

Efficient Adjustments & Corrections

Inventory ledgers make it easy to adjust weights and counts to resolve discrepancies. AirMed ensures that backdated adjustments are logically aligned with all prior and subsequent records. Adjustments that affect inventory levels are clearly categorized as ‘other additions’ or ‘other reductions’ in monthly compliance reports.
Error Correction Options

In the event of data entry errors, AirMed offers multiple correction tools. For example, if an inventory item is created with the wrong date, admin tools can be used to correct the creation date to match the creation of the parent record (e.g., adjusting a lot’s creation date to align with the date of the harvest). All changes are logged in both the affected item’s ledger and the parent record’s ledger. Additionally, new options are available to delete incorrect records or merge materials back into their original source when appropriate.
The benefits of our new inventory ledgers include more accurate tracking, improved operational efficiency and easier compliance. For more information about AirMed 5 visit our Software page.
Production Orders in AirMed 5
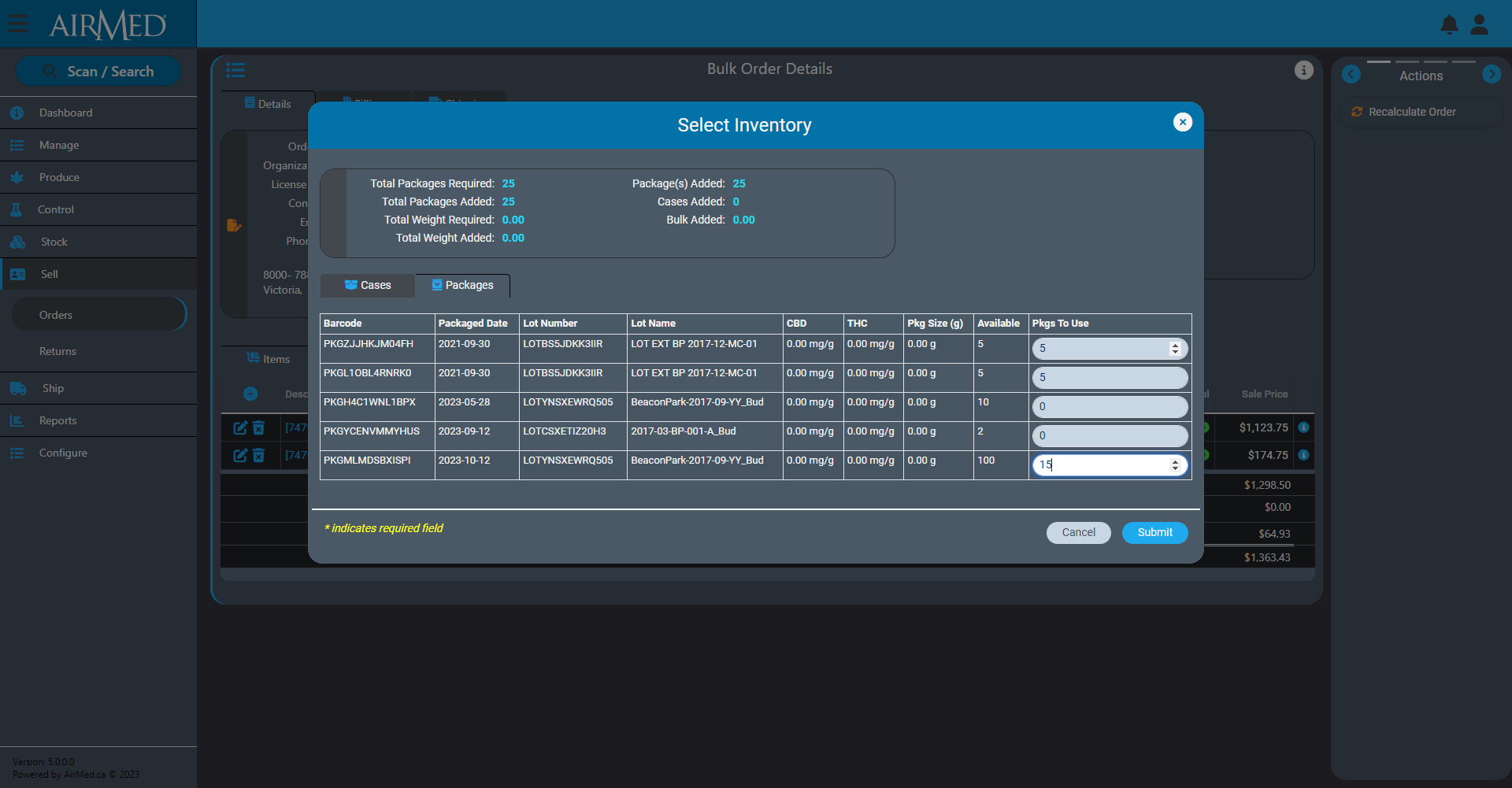
Our new Production Orders features will offer the ability to synchronize AirMed more easily with external enterprise resource planning (ERP) systems such as NetSuite or Microsoft Business Central.
When combined with tasks and approvals, this provides a high level of automation and ensures that product that is created meets market demand.
Internal Production Orders
Production Orders are essentially a wrapper for activities related to producing material such as cannabis, extractions, packages, topicals, or edibles.
Internal Production Orders are used to plan for production over a specified period of time. For instance, if producers are involved in cultivation, they can create internal production orders to produce new batches, harvest and dry them, and produce bulk products for sale or for additional processing.
Once a production order is created, it can optionally cascade out all the tasks related to the item being produced, even if some of the production requires routing to external organizations and suppliers.
Internal production orders can be used to plan production for any period of time. Multiple internal production orders can cover an entire year for a producer.
External Production Orders
External production orders are based on a sale to another organization, such as a provincial government distribution warehouse. An external production order would typically be tied to a purchase order from an external organization.
Using this feature, a producer will be able to see if there is existing inventory to fill an order. A producer can also create a new order that will require either growing and producing new cannabis or sourcing from another licensed producer.
Once the production order is approved (using the new Approval Workflow in AirMed), it will cascade out all related tasks to complete the order.
For instance, a producer makes a sale to a provincial distribution warehouse for 2000 vape cartridges. The producer can create a new batch of plants as source material and then track the entire process from propagation to finished packages in master-cases loaded on a pallet to be shipped.
The entire production process can be tracked and reported on, even if the workflows to produce the vape cartridges require a third-party to manage the extraction and packaging stages.
For more information call or email us, fill out the request demo contact form or visit our Software page.
Introducing Our Updated WordPress Medical Plugin
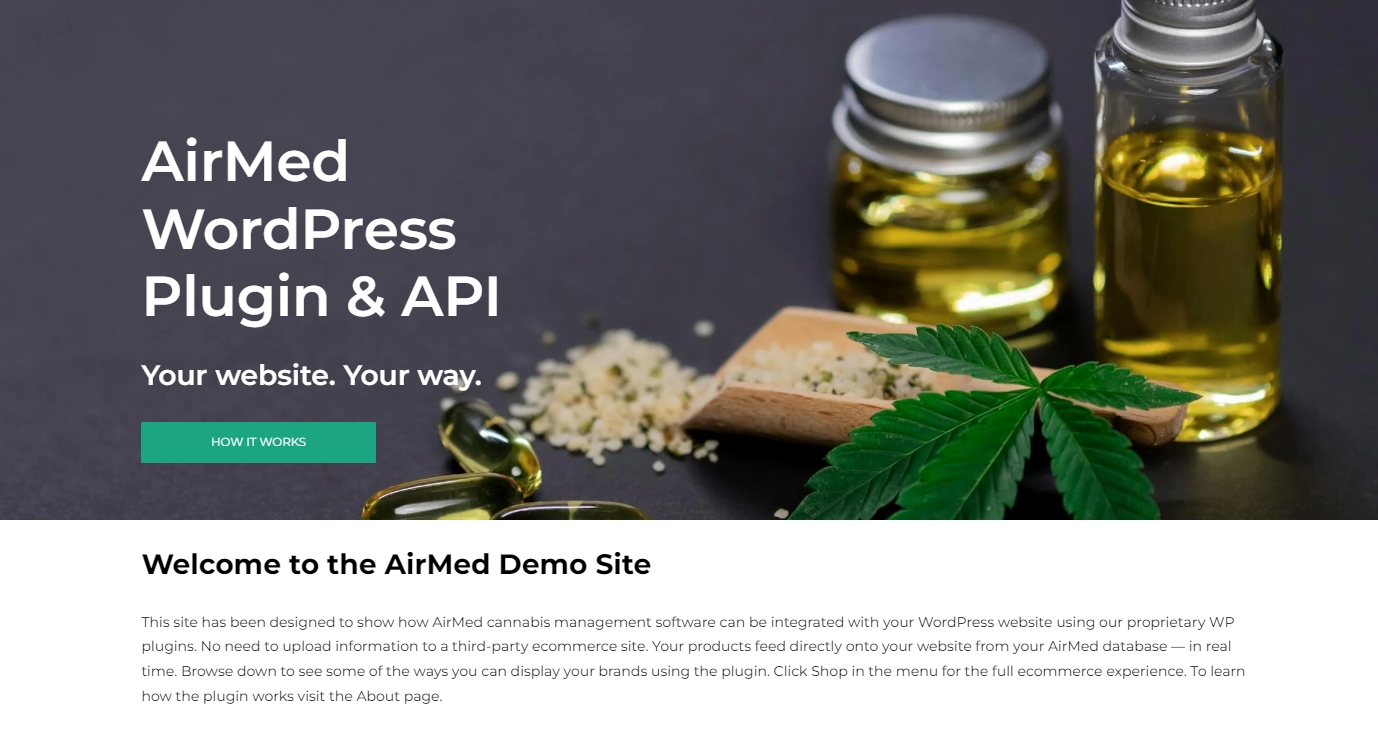
Elevate Your Medical Cannabis Website Experience
We’re excited to announce the release of our enhanced WordPress Medical Plugin — now better than ever to help you build a cutting-edge medical cannabis website in Canada.
The AirMed Medical Plugin seamlessly integrates with Wordpress websites, letting licensed Canadian cannabis producers launch a fully functional medical cannabis platform. No need to upload product information to a third-party ecommerce site. Your brands and products feed directly onto your website from your AirMed database — in real time.
Our latest update, driven by customer feedback, introduces powerful new features designed to make your website stand out in a competitive market.
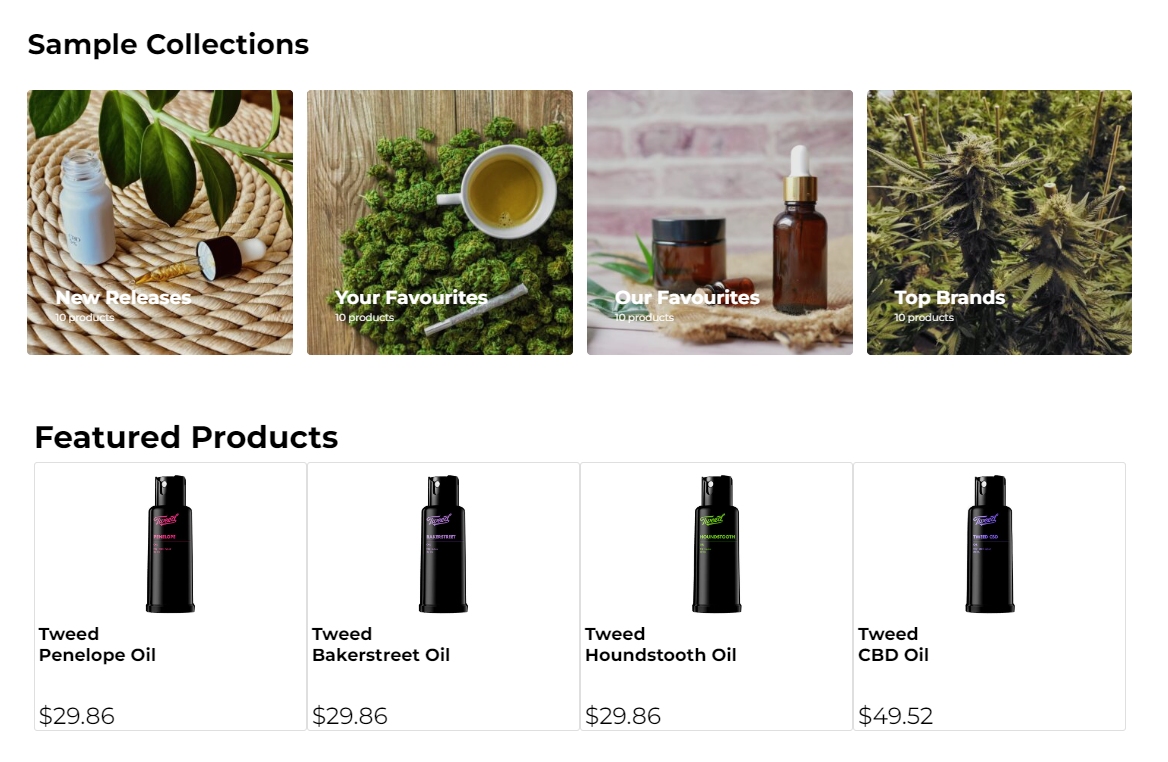
What’s New?
- Enhanced User Experience: We’ve added a sleek new carousel control, making it easier to showcase your products and services in a visually engaging way. Paired with optimized metatags, your site will not only look better but also attract more traffic through improved search engine visibility.
- Mobile-Friendly Design: Recognizing the importance of mobile users, we’ve updated all in-built webpages to deliver a seamless experience on smartphones and tablets. Your patients can now access crucial information, no matter where they are, with the same level of quality as on a desktop.
- Dynamic Dashboard Widgets: Stay on top of your site’s performance with our new dashboard widgets for summary pages. Get insights at a glance, making it easier than ever to manage and optimize your content.
With our plugin, patients can register, submit applications, and purchase medical cannabis — with all data securely encrypted in full compliance of Health Canada regulations.
As an AirMed customer, you can quickly install the plugin and add a simple WordPress shortcode to display your product catalog.
Whether you’re a clinic or medical cannabis producer, our updated WordPress Medical Plugin gives you the tools you need to create a compelling, user-friendly online presence.
Get started today and take your medical cannabis business to the next level!
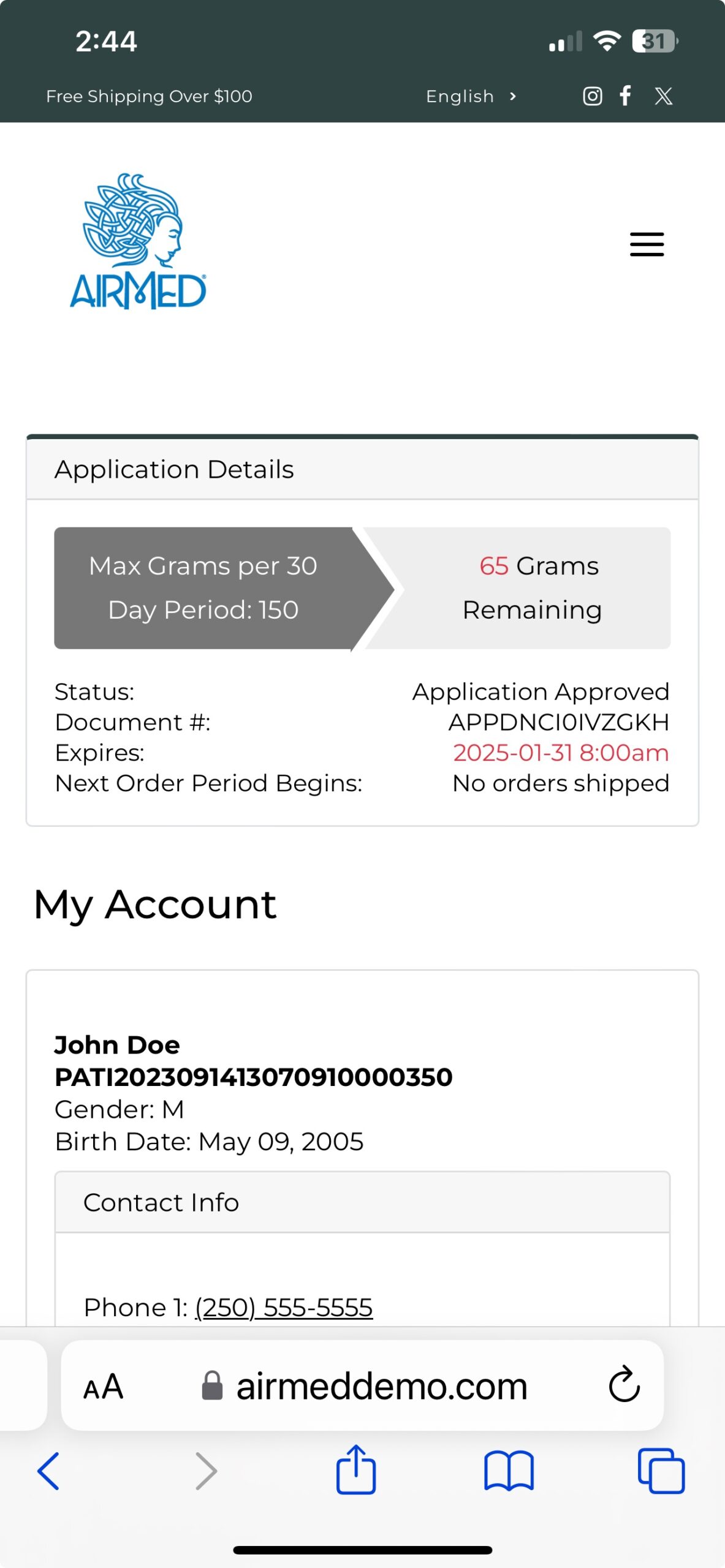
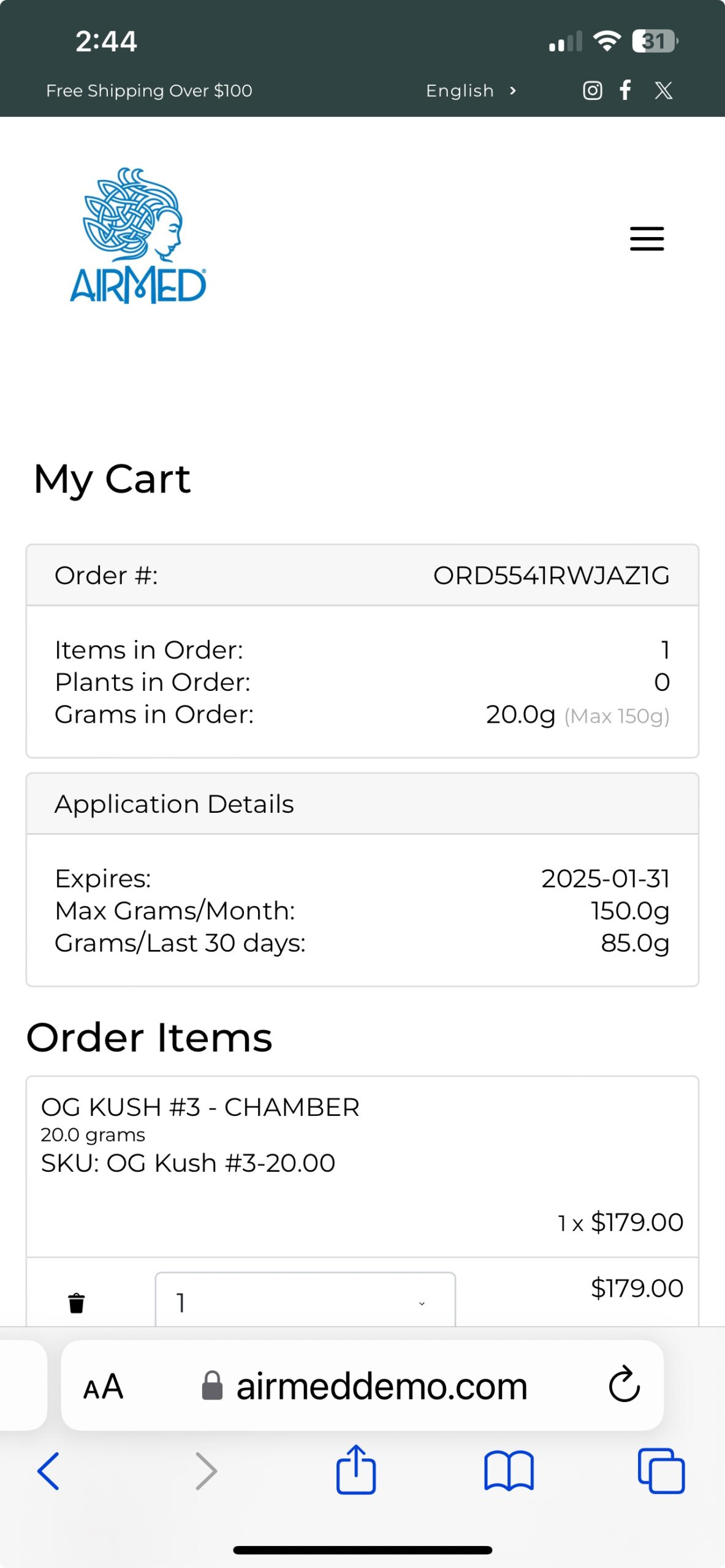
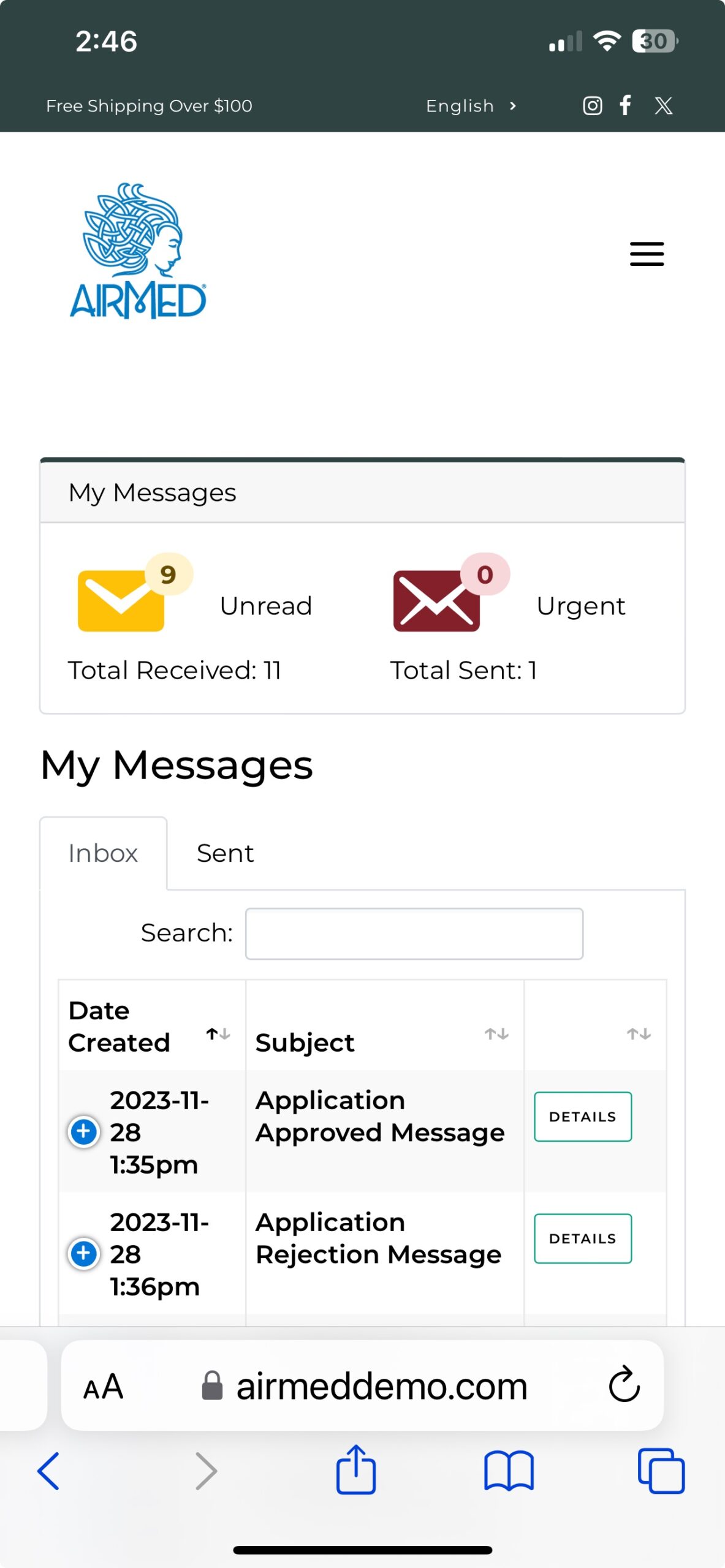
Call us toll-free at 1-877-313-2442 to learn how our plugin can help you achieve your goals in the medical cannabis marketplace.
To see our plugin in action, visit our live demo site at airmeddemo.com.
AirMed Supports Direct Delivery
What is Direct Delivery?
Direct delivery programs let licensed producers sell their products directly to retailers, which can eliminate warehousing. In British Columbia, the standard practice is for LPs to send cannabis to the BC Liquor Distribution Branch’s (LDB) warehouse. Dispensaries must then choose the products they wish to buy from the LDB product list.
For retailers, the disadvantages include limiting choices to what’s available in the warehouse and typically requiring a minimum order amount. This process also does not allow retailers to select products based on freshness.
For LPs, there is no assurance that the cannabis sent to the warehouse will be ordered within its shelf life. Yet each time a product is shipped, the production facility must use a pre-paid excise stamp. As a result, LPs spend the price of the excise stamp without a guarantee of sale. The possibility exists for the cost of producing and shipping the product to be thrown away, plus there’s the extra work involved in reclaiming the excise tax.
Direct delivery lets LPs register and list products for sale through a provincial direct-to-retailer program. Dispensaries can choose products from an individual LP and arrange for delivery directly from production facility to storefront. The shipment still requires an excise stamp and provincial fees, but the sale to the retailer has already been made, which covers those costs.
The benefits of direct delivery include:
- Giving retailers greater choice when ordering plus no minimum sales and potentially shorter delivery times
- Supplying dispensaries and consumers with fresher product
- Letting LPs cultivate-to-order and preventing products from expiring in warehouses
- Removing speculative costs of excise stamps for LPs and ensuring up-front revenue to LPs
- Helping small-scale producers be more competitive in the marketplace by creating opportunities for brand marketing and promoting better customer service to retailers
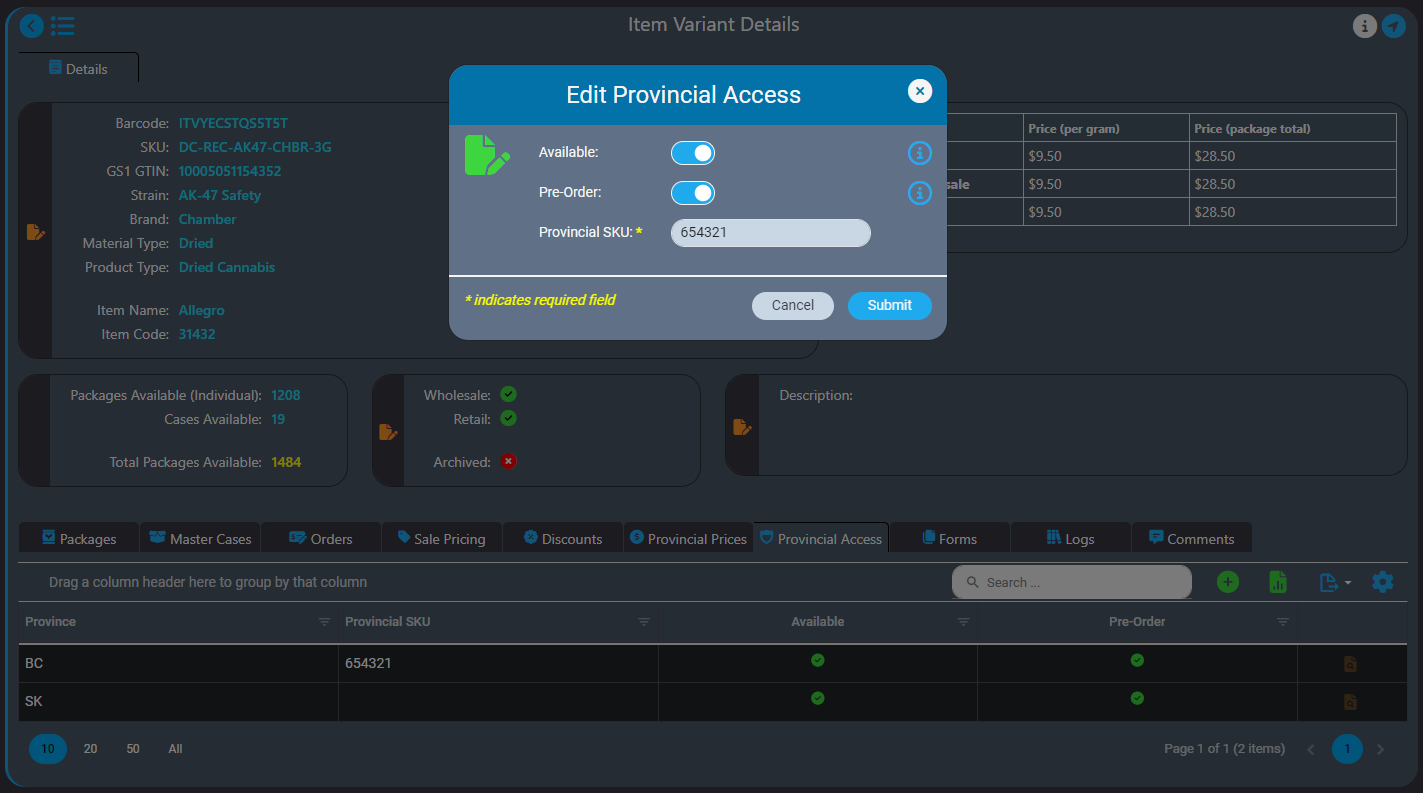
How AirMed Supports Direct Delivery
AirMed Direct Delivery offers a streamlined service that enables licensed retail stores to bypass provincial warehouses and order products directly from licensed producers who use AirMed in provinces where this is permitted, currently British Columbia and Saskatchewan.
Our built-in workflows that support direct-delivery simplify your processes making it easier for you to work with your retail partners.
WordPress Plugin for Retailer Registration
A proprietary WordPress plugin and API provide fast and easy integration of your AirMed database with an online store on your website. This lets you offer your products directly to licensed retail outlets complete with a catalog and shopping cart. Retailers can register and get approval from licensed producers through the WordPress plugin. Approved retailers gain access to a product catalogue and order cases of finished packages.
Pricing and Orders
AirMed supports different pricing structures for various provinces for both individual packages and bulk cases. AirMed also allows pre-ordering of out-of-stock products, enabling producers to manufacture based on incoming orders.
Order Fulfillment
Once an order is placed, it is automatically processed in AirMed. Producers can fulfill orders, manage payments, and ship products directly to retail stores.
Provincial Support
Direct delivery is designed to comply with current provincial regulations and can adapt to include new provinces as regulations change. You can even set up direct-delivery product availability for provinces that may elect to allow direct delivery in the future.
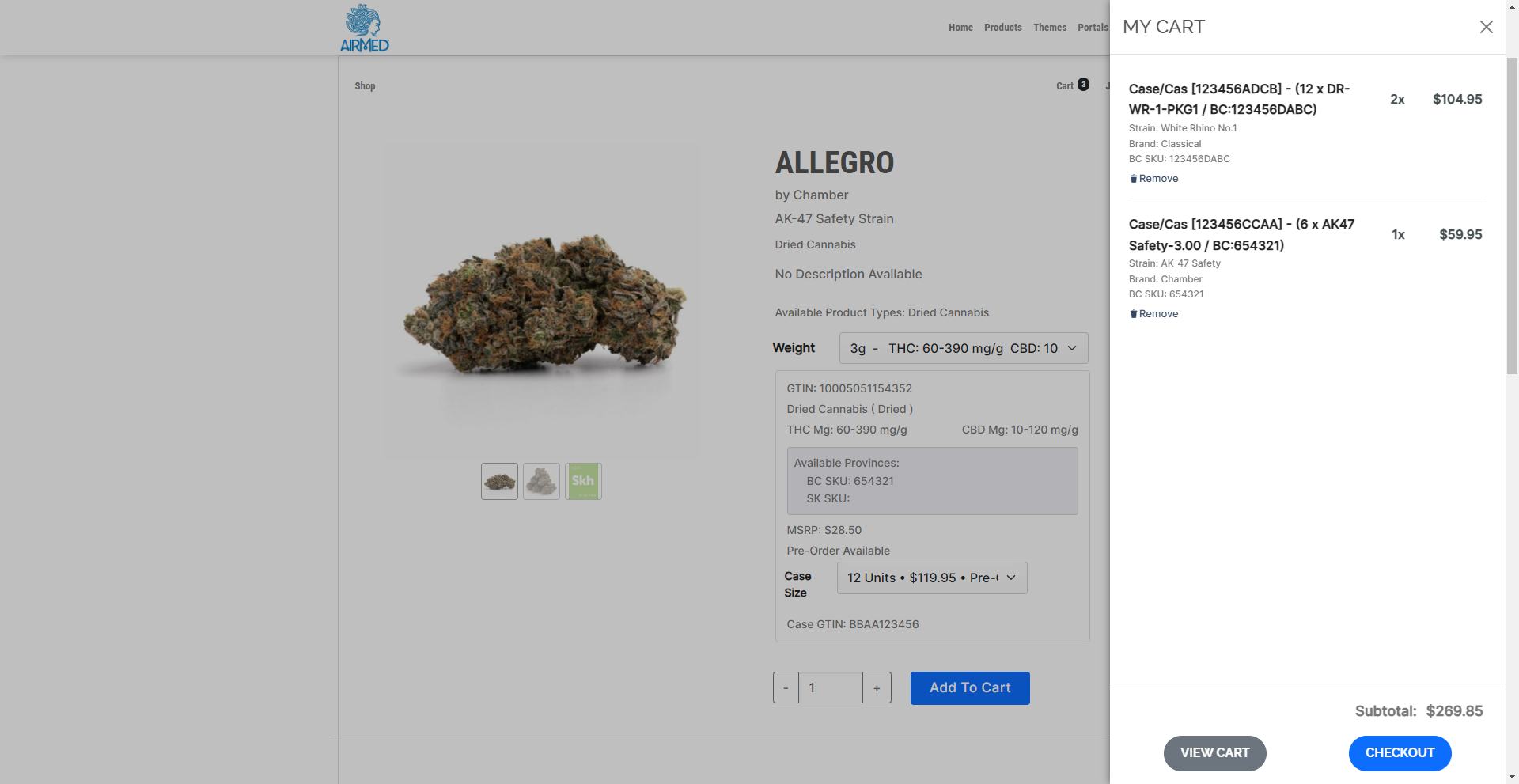
Our solution is ideal for those looking to streamline their supply chain and enhance their ordering process in the cannabis retail industry.
Efficiency: Retail stores can directly access products from producers who use AirMed, reducing the dependency on provincial warehouses and potentially shortening delivery times.
Flexibility: Producers can manage their inventory and production schedules more effectively through pre-orders, and AirMed manages creating product cases in varying sizes with associated case pricing for each product type and province.
Scalability: Our system can expand to include additional provinces as they permit direct delivery.
To see all the ways AirMed supports direct delivery, book a free demo by using the Request Demo button at the top of the page.
For more information on direct delivery in British Columbia, visit: https://www.bcldbcannabisupdates.com/LDBDirectDeliveryProgram
For more information on cannabis laws in Saskatchewan, visit: https://www.slga.com/cannabis
To read an article about direct delivery on the Stratcann website visit: https://stratcann.com/insight/bcs-
annabis-direct-delivery-program-is-growing-but-fees-still-too-high/c
Selling internationally? You need GS1 and GTIN!

GTIN or Global Trade Item Number is a standard from the GS1 (Global Standards) organization. GTIN consists of unique codes that identify manufacturers and their products using barcodes. When scanned by an electronic reader, the GTIN barcode provides a code that is related to a specific manufacturer and a specific product from that manufacturer.
In North American, the UPC (Universal Product Code) is an existing form of the GTIN. In Europe, EAN-13 is the GTIN standard.
The GTIN system lets you and your products be identified across the globe. If you hope to sell in certain parts of the world, such as Europe, you will need to use GS1 standards and GTIN codes. Once you have signed up with your regional GS1 office, you will be issued a series of unique codes to use on your product packaging.
Fully supporting GS1, AirMed prints your GTIN barcodes directly from the database. Our master case processing lets you use multi-level barcoding for several layers of packaging or stock-keeping units (SKU). For example, one case could contain a dozen smaller cartons. Each of those cartons could contain a dozen retail-ready individual packages. All of those packaging layers can have its own barcode to meet the retail standards of the region where it will eventually be sold.
AirMed helps you meet your barcoding and packaging standards whether selling to a provincial warehouse or shipping internationally. AirMed even lets you apply pricing at the package or the master case SKU, and custom pricing can be set for specific SKUs by customer.
For more information on GS1 standards and GTIN barcoding, visit: https://gs1ca.org/
If you’d like to discuss your specific needs, please give us a call at 1-877-313-2442 or click the Request Demo button at the top of the page to start the ball rolling.
Data collection made easy with AirMed e-form designer
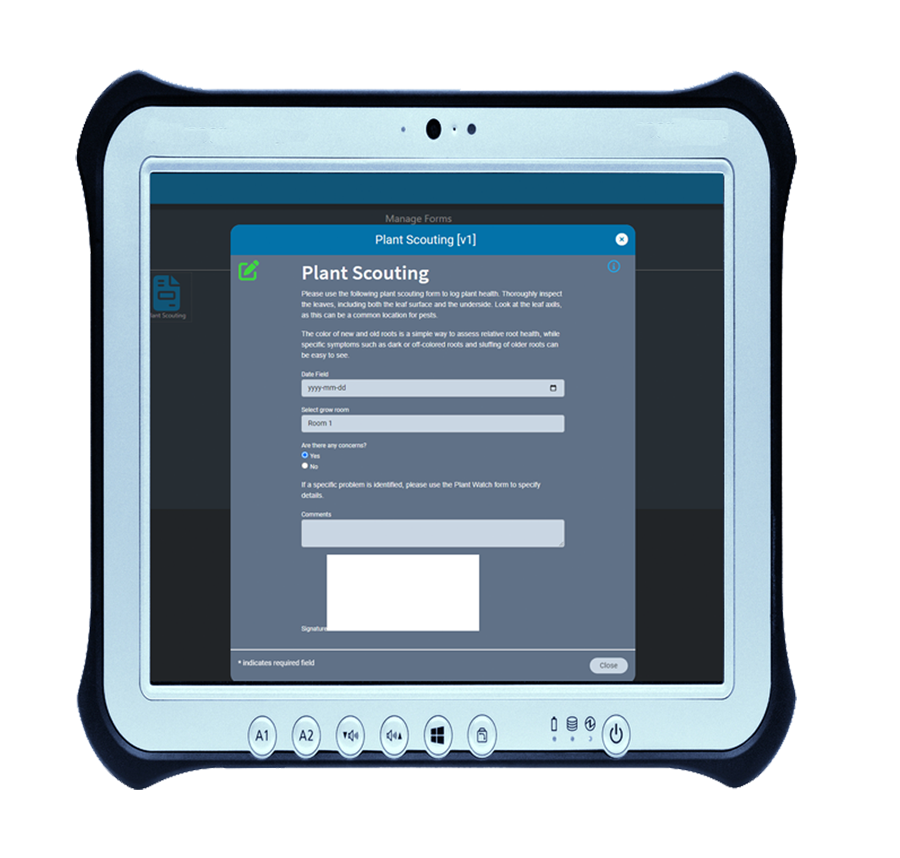
For many compliance-based businesses, forms are unavoidable. Even with the most comprehensive software, forms are sometimes necessary for collection and verification of data.
While many software vendors offer electronic form functionality, in most cases the information contained in those forms is not stored in the database. This means relying on PDFs or printed documents to access the data contained in the form.
Our built-in form designer lets you create custom electronic forms that integrate with AirMed’s system data-sources. All fields in electronic forms created using the form designer can be stored in the AirMed database. There is no need to refer to physical documents or PDFs.
Using AirMed, you can automate workflows using custom e-forms to prevent the need for paper-based documentation. Forms can be linked to processes such as plant scouting, nutrient checks, room sanitation, and shop floor data collection. Using our form designer, you can employ drop-down picklists, date & time, checkboxes, radio groups, custom fields and more. AirMed lets you apply auto-complete, file upload, hidden input and electronic signatures to your forms.
All the data is electronically accessible and even reportable.
Form data collection doesn’t get any easier than that.
For more information visit our AirMed 5 page.
Workforce Management in AirMed 5
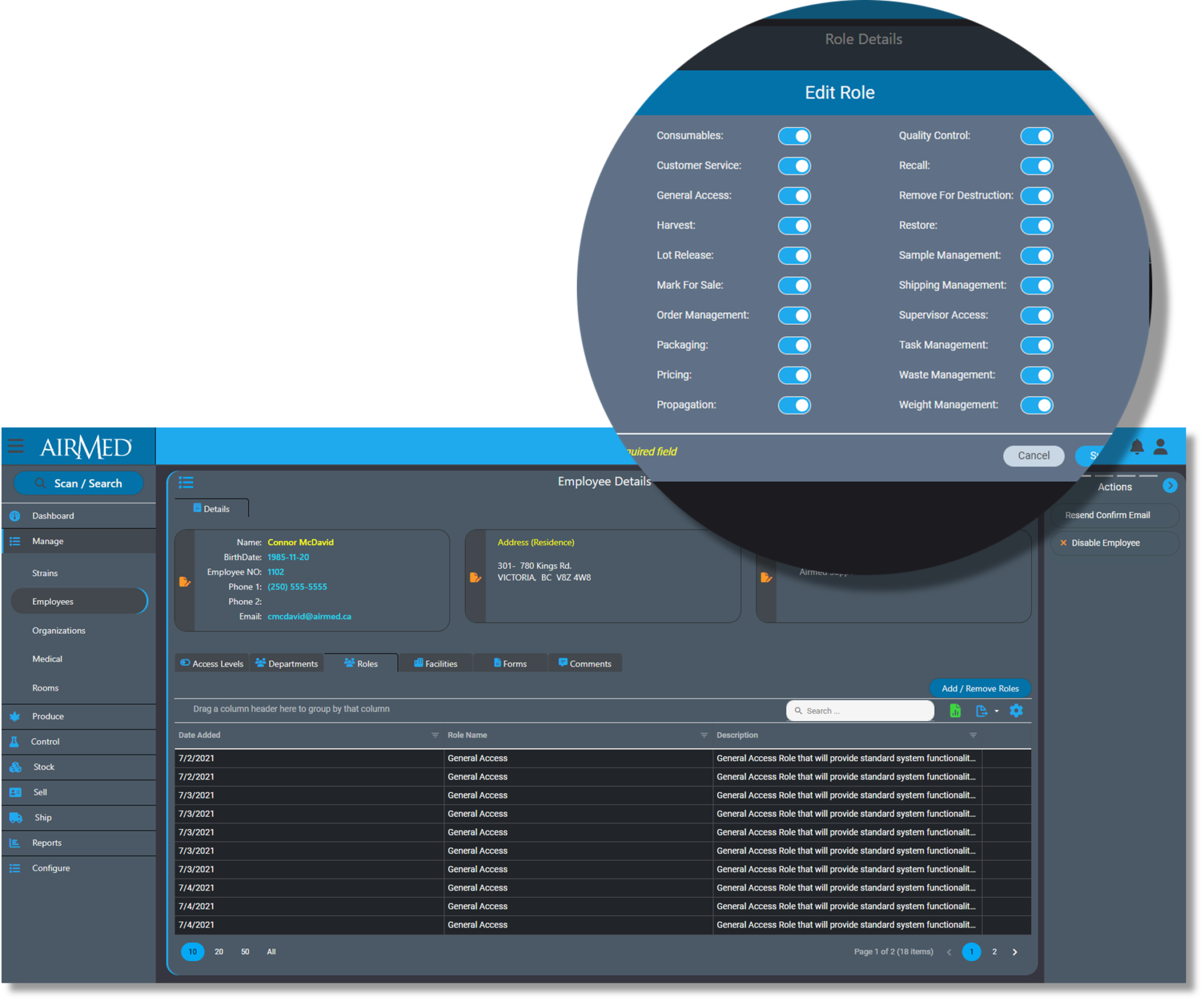
AirMed simplifies compliance by tracking and logging every action and ensures audit readiness with reports and analytics.
Employees only have access to functions and data that relates to their job responsibilities. System Administrators can only access data through approved computers utilizing a VPN connection.
And AirMed offers licensing by facility size rather than by individual user to encourage producers to configure individual accounts for every worker.
Our system improves quality management by allowing producers to identify gaps in employee knowledge and skills.
To see for yourself what AirMed can do, click the Request Demo button at the top of the page.
For more information visit our Software page.
Implementing AirMed in Your Facility

As a cloud-based platform, AirMed does not require expensive dedicated hardware to operate. Producers are free to use any computing device that supports a web browser. As a result, set up consists mainly of configuring the software to meet your needs. The rest of the time involves learning to use the system and getting your employees up and running.
How AirMed is implemented within your organization depends on the areas of the software you’ll be using. The first step in any software implementation is a needs assessment to help identify exactly how the software will be used. Understanding the capabilities of AirMed and how it can work with your processes is one of the keys to successful implementation. Your AirMed implementation specialist will work with you to perform an assessment of your needs to determine which areas of the software should be configured.
We provide an implementation guide and a production checklist that you can go through with your AirMed Implementation Specialist. Together, you’ll review the functionality in AirMed and determine which areas and functions are needed for your business operations. AirMed includes the ability to disable navigation menus for areas that you won’t be utilizing to provide a streamlined interface for your workers.
When you have completed your assessment and training, the next step is to configure your Live Production environment. After you’ve set up and tested your system, it’s time to train other users (your workers) to use AirMed.
Once users have usernames and passwords, they can access the training resources in the AirMed Learn environment. Workers can take additional training at a later date to develop skills for different areas of the system — in fact, anyone with access to AirMed can use the Learn environment at any time to practice new workflows or refresh their knowledge. When employees have completed training, you can give them access to the system, so they can get to work.
With AirMed you can implement only the functionality you need right now. For example, you can start by implementing the AirMed Grow module, then when you expand your operation, you add modules that meet your current needs for performing extractions, packaging for provincial sales, selling to medical patients or whatever your business entails.
Working with your implementation specialist will help you see all the benefits of using AirMed and ensure that you are getting the most from our software.

For more information on how AirMed helps specific types of businesses, visit our Customers page or our Frequently Asked Questions page.
Ready to learn more about AirMed? Click the Request Demo button or call 1-877-313-2442.